An eCOA/ePRO solution should unleash new ways to engage patients, strengthen retention, improve data quality and reporting, and support better outcomes.. But you are unlikely to realize those results with a solution that simply digitizes paper processes. What does it take to design and implement a solution that delivers modern digital experiences for patients? Look for these must-have capabilities.
Capability #1: All-in-one configuration
A one-size-fits-all platform will fall short of addressing the unique needs of your trial. But a fully custom solution will almost certainly exceed your timeline and budget. To clarify, look for a solution that splits the difference. Thus offering a set of core functionality that can be configured based on your needs.
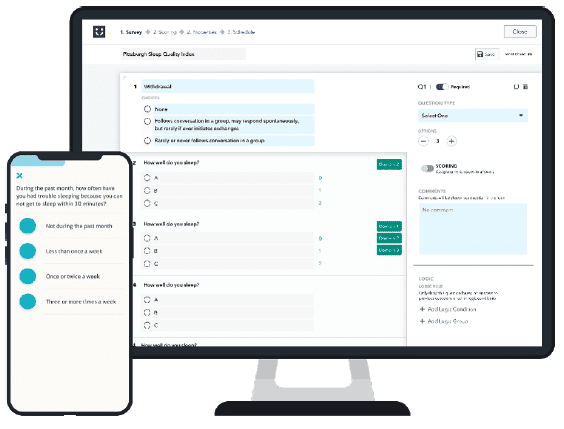
Ideally, it should be possible to design your solution through a single interface and deploy it through a single solution profile and platform. Configuration should address all the functionality you need: engagement, ePRO, virtual visits, consent and more. For patients, all functionality should be available through a single app. Most importantly, a platform should be able to support design and deployment in hours or days, not weeks or months.
Capability #2: Device flexibility
The Bring Your Own Device (BYOD) model has emerged as a hot topic in the clinical trial industry. Earlier conversations about BYOD prompted questions about being able to deploy validated questionnaires. But as COVID-19 pandemic shutdowns have prompted greater adoption of electronic solutions, even the holdouts now see the value in BYOD.
BYOD support — for both Apple and Android devices — is a must in a modern eCOA/ePRO platform. Look for a solution that empowers patients to use their own smartphones to do everything from completing eConsent and survey instruments to engaging with your communications and content from their own device.
Of course, BYOD may not be the right approach for every patient in every trial. In some cases, BYOD will work for most, but some patients will still need the option to use a provisioned device. In other cases, a trial may need to provide web-based access to the eCOA/ ePRO solution. A modern platform should offer the flexibility to accommodate BYOD, provisioning, and web — in whatever combination your trial and your patients require.
Capability #3: Self-service patient enrollment
We no longer need to visit a bank branch or clothing store to create an account with those businesses. Similarly, patients should not have to visit an investigative site to get started in a study. Look for a solution that supports self-service enrollment. If you have identified your patients — and are open to using a BYOD model — this approach reduces or eliminates the need for an in-person visit.
It should be fast and easy for patients to visit an app store, download your ePRO/eCOA app, and create a login with two-factor authentication. For patients with provisioned devices, you should be able to preload the app and provide login credentials via email. If it makes sense for your trial, it should also be possible to incorporate eConsent into this onboarding workflow.
Capability #4: Passive data collection
One of the greatest advantages of a modern and engaging eCOA/ePRO solution is proximity to the patients in a trial. A mobile app gives voice to patients on how their treatment affects their quality of life. Seek a platform that provides multiple channels for passive data collection. You should be able to collect data via the app at pre-determined intervals, on demand, or continuously. There should be mechanisms for collecting data at both scheduled and unscheduled visits. And there should be ways of “checking in” without directly disturbing a patient. Examples include connected wearables, tracking of screen time and/or steps taken, and general use of the patient mobile app.
Capability #5: Timely insights
With paper-based processes, there are understandable delays in access to study data. When you transition to electronic outcomes reporting, you should expect much faster access to data.

A platform should incorporate principles of modern data science to serve up “news you can use.” These snapshots should include insights into whether the compound you are investigating is safe and effective, as well as insights into the demographics, enrollment status, and compliance status of all patients in the study. Expect the ability to drill down by patient segments or individual patients, so you can proactively identify and address at-risk subjects before they drop out of the study.
About the Datacubed platform
The Datacubed platform has been designed to be flexible, easy to modify, and quick to deploy. Although clinical trials have become more complex, managing them should be simple. With the Datacubed platform, you can set up and launch your study in hours, not weeks. You can control multiple studies simultaneously, but each study has its own participant, instrument management, and data monitoring in real-time. Check out this short demo video to see the platform in action!

